
AAOS 2016: Synthetic hydrogel implant vs. MTP arthrodesis for hallux rigidus

AAOS 2016: Synthetic hydrogel implant vs. MTP arthrodesis for hallux rigidus
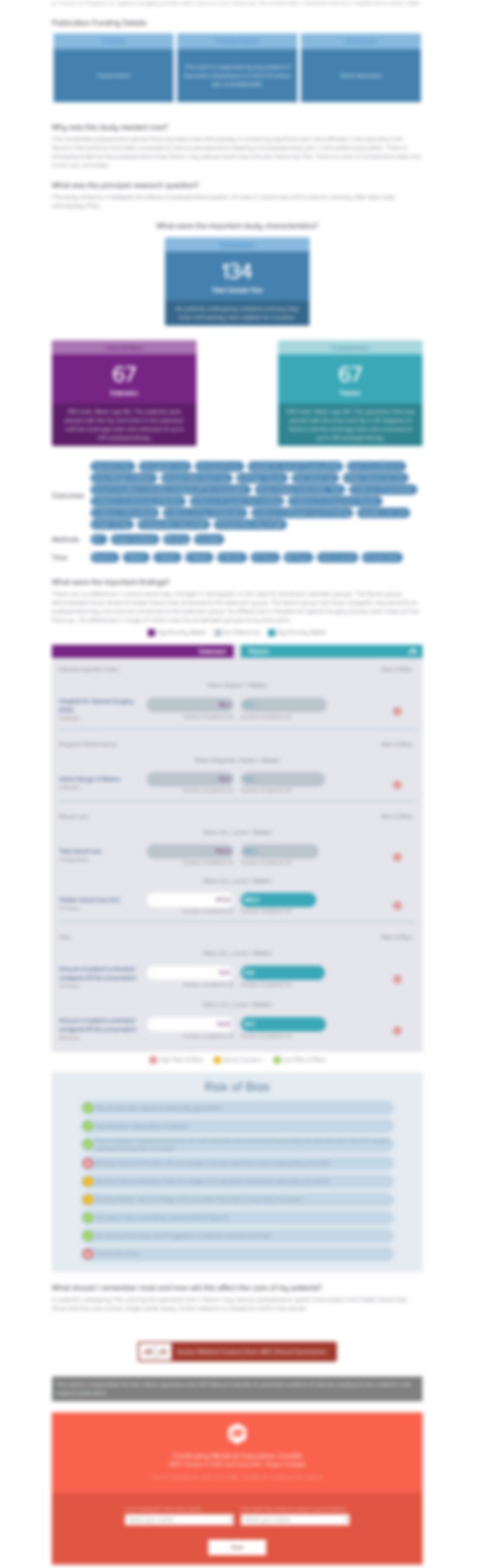
Prospective Randomized Multi-Centered Trial Comparing a Synthetic Implant to 1st MTP Fusion in Hallux Rigidus
Did you know you're eligible to earn 0.5 CME credits for reading this report? Click Here
CONFERENCE ACE REPORTS
This ACE Report is a summary of a conference presentation or abstract. The information provided has limited the ability to provide an accurate assessment of the risk of bias or the overall quality. Please interpret the results with caution as trials may be in progress and select results may have been presented.
Synopsis
202 patients with advanced stage hallux rigidus were randomized to one of two treatments: synthetic hydrogel cartilage implant or standard first metatarsophalangeal (MTP) arthrodesis (joint fusion). The purpose of this study was to evaluate pain and function (active dorsiflexion motion) in comparison to one another, in addition to recording adverse events and requirement of secondary surgeries. Re...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.